DSCSA Serialization Requirements
Download HDA’s 2022 Serialization Readiness Survey
On November 27, 2018, the DSCSA required all trading partners to serialize product with a unique product identifier (UPI). If unit dose is supplied, Serialized Numerical Identifiers (SNI) and Unique Product Identifiers (UPI) are required to be on each sellable unit down to the unit dose level.
A product identifier is defined as a “standardized graphic” including both human and machine-readable data that contains the standardized numerical identifier, lot number, and expiration date of the product see the HealthFirst SNI labeling advantage.
Trading partners are also required to exchange product tracing information when transacting specific prescription drugs. Tracing requirements established under section 505D of the FD&C Act require trading partners capture and maintain the applicable tracing information for no less than 6 years post transaction date and be able to provide that information when requested by the FDA or other Federal or State official in the event of recall or for the purpose of investigating a suspect or illegitimate product. The product tracing standards include Transaction Information, Transaction History, and Transaction Statement. In 2023, this new requirement will apply to dispensers and pharmacies as well.
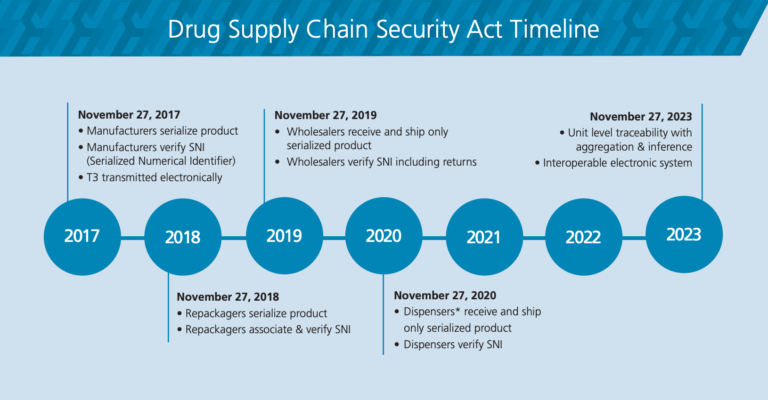
Implementing serialization of single or LUM items in compliance with the phases of the DSCSA regulation is complex, costly, and labor-intensive. Considerations include State and Federal Repackager licensing and regulatory compliance, production process investments, label and packaging design, labelling and repackaging production time. HealthFirst is proud to say that we were in compliance prior to the November 2018 deadline.
For more information on the DSCSA requirements and HealthFirst compliant distribution, download the Overview of Pharmaceutical Serialization Compliance and DSCSA guide below.
Contact Us
Contact us to discuss your DSCSA compliant distribution needs.
